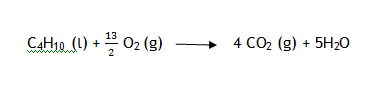Enthalpy of combustion
Heat released when 1 mole of substance is completely combusted in oxygen gasex:
Enthalpy of neutralization
Heat released when 1 mole of water is formed when acid reacts with base
ex:

Enthalpy of hydration
Heat released when 1 mole of gaseous ion is hydrated in aqueous solution
ex:

Enthalpy of formation
Heat change when 1 mole of compound is produced from its element
ex:

Enthalpy of solution
Heat change when 1 mole of substance dissolves in solvent/water (dissolution) to form infinite dilute solution
ex:

Enthalpy of atomization
Heat absorbed when 1 mole of gaseous atom is formed from its element
ex:
Enthalpy of sublimation
Heat absorbed when 1 mole of substance in solid state is sublimed
ex: